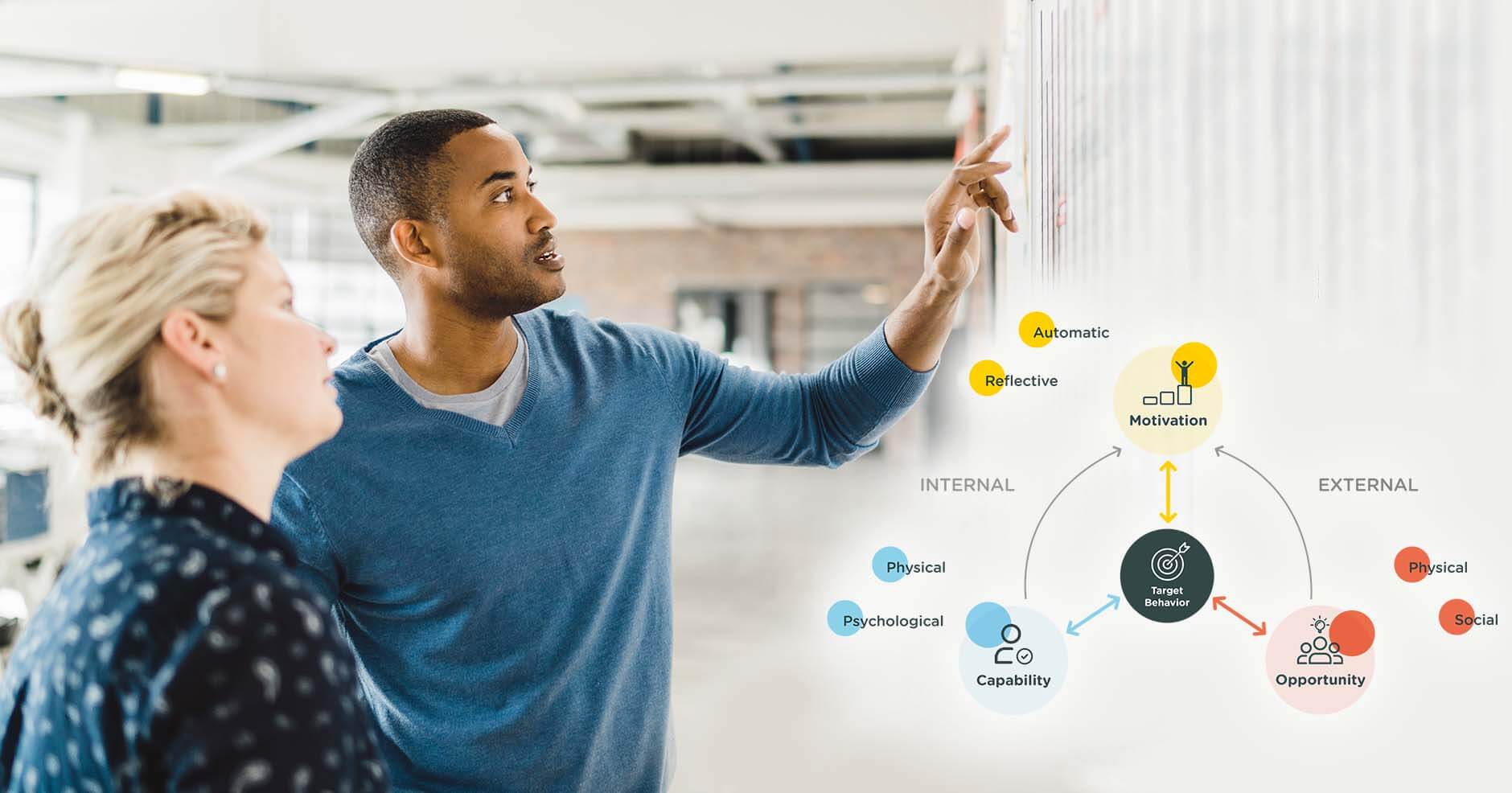
We help life science companies to define and execute their digital health strategy, with a focus on transforming patient experiences and outcomes within their therapeutic area of interest.
For a digital health solution to have a real impact, it must deliver on the needs of all stakeholders to secure successful launch, integration into care delivery pathways, adoption by its intended end-users, and return for the company sponsor. This requires a rigorous and holistic approach from the start.